Two Epidemics, One Root Cause. Are We Just Beginning to See the Connection?
- Metabolic Mind Project

- Apr 11
- 8 min read
The United States is currently managing two public health crises that have been building in parallel for decades. One is metabolic. The other is psychiatric. Neither shows signs of reversing. And for the most part, medicine has treated them as separate problems with separate solutions.
They are not separate. The evidence linking metabolic dysfunction to psychiatric illness has been accumulating for years, but it has not yet crossed over into the mainstream conversation about mental health. Understanding that connection, what it means, why it exists, and what it implies, is one of the most important things anyone thinking seriously about mental health can do right now.
This article introduces that connection. It is deliberately broad, because the relationship between metabolic and mental health touches everything from obesity to diabetes to dementia, and no single article can do justice to every thread. Consider this a map of the territory. Future articles will go deeper into each region.
Two epidemics. One metabolic. One psychiatric. Both worsening. Both largely treated as unrelated. The evidence says otherwise.
The metabolic crisis in numbers
The scale of metabolic dysfunction in the United States is difficult to fully absorb. More than 42% of American adults now have obesity, a figure that has more than doubled since the 1970s [1]. Approximately 38 million Americans have diabetes, with Type 2 diabetes accounting for the overwhelming majority. An additional 96 million, roughly one in three American adults, have prediabetes, most of them undiagnosed [2].
These numbers represent more than physical health consequences. They represent tens of millions of people living with biological conditions, specifically insulin resistance, chronic inflammation, mitochondrial stress, and dysregulated blood sugar, that directly affect how the brain functions. The downstream effects on mood, cognition, and psychiatric vulnerability are not incidental. They are mechanistic.
Insulin resistance is the common thread running through obesity, Type 2 diabetes, and metabolic syndrome. When cells stop responding properly to insulin, the consequences ripple outward through virtually every organ system, including the brain. The brain is not a bystander in metabolic dysfunction. It is one of its primary targets.

The parallel psychiatric crisis
At the same time that metabolic dysfunction has been rising, psychiatric medication use has been escalating at a pace that is difficult to explain through increased awareness or improved diagnosis alone. Antidepressant prescribing in the United States rose nearly 400% between 1988 and the mid-2000s, making antidepressants the third most prescribed class of drugs for adults aged 18 to 44 [3]. As of 2023, over 59 million American adults received mental health treatment or counseling, with prescription medication being the most common form of intervention [4].
These trends have unfolded alongside the metabolic crisis, not after it, not before it, and not independently of it. The overlap is not coincidental. Obesity is associated with significantly increased lifetime rates of major depression, bipolar disorder, and panic disorder [5]. People living with metabolic syndrome are at substantially elevated risk for anxiety and depression. And the relationship runs in both directions: psychiatric conditions worsen metabolic outcomes, and metabolic dysfunction worsens psychiatric ones.
The conventional response to this pattern has been to treat the psychiatric symptoms pharmacologically, often with medications that themselves contribute to weight gain and metabolic dysregulation, potentially deepening the underlying problem. This is a cycle that medicine is only beginning to acknowledge.
Antidepressant prescribing rose nearly 400% in two decades. In the same period, obesity rates more than doubled. These trends did not unfold in parallel by coincidence.
Diabetes and mental illness: the numbers you need to know
The relationship between diabetes and psychiatric illness is among the best-documented in medicine, and among the most consistently ignored in clinical practice.
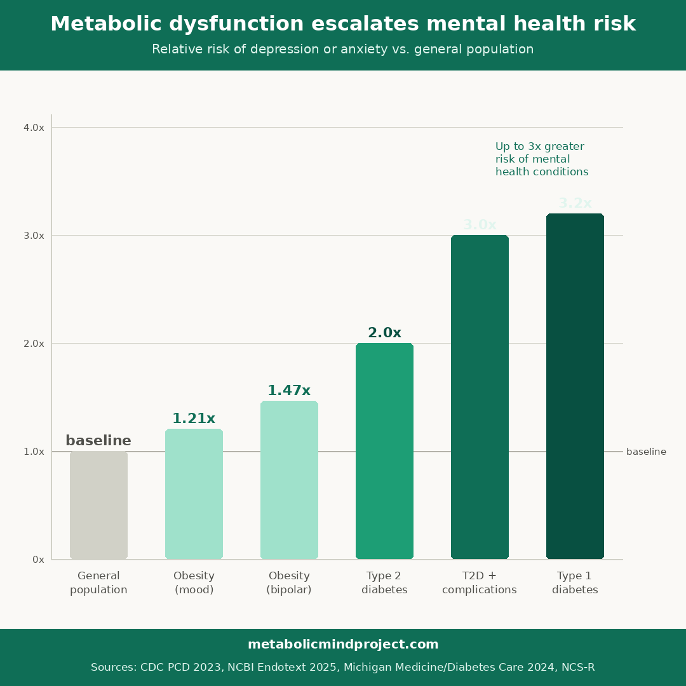
Depression prevalence is approximately doubled in people with Type 2 diabetes compared to the general population [6]. For Type 1 diabetes, the figure is more striking still: a systematic review found depression prevalence to be more than three times higher in Type 1 diabetics than in non-diabetic adults [7]. For people with diabetic complications, a large Danish cohort study published in Diabetes Medicine found that complications raised depression and anxiety risk with a hazard ratio of 1.77, with amputation showing the strongest individual association [8].
A 2024 study published in Diabetes Care found that people with chronic diabetes complications had up to three times greater risk of having a mental health condition such as anxiety or depression, and that this effect increased as adults got older [9]. The relationship is bidirectional: those with mental health disorders were up to 2.5 times more likely to experience sustained diabetes complications. Each condition actively worsens the other through shared biological mechanisms, including inflammation, HPA axis dysregulation, and disrupted glucose metabolism.
Anxiety is also more prevalent in people with diabetes. The CDC reports that people with diabetes are 20% more likely than those without to have anxiety, and a systematic review found elevated generalized anxiety disorder prevalence across multiple populations with diabetes [10]. Among people with diabetes, depression increases the risk of mortality by approximately 50% [6].
These are not small effect sizes. They represent a population of tens of millions of Americans living with both a metabolic condition and a significantly elevated psychiatric risk, in a healthcare system that rarely screens for both simultaneously.
What is actually happening biologically
The connection between metabolic dysfunction and psychiatric illness is not simply about the stress of managing a chronic disease, though that is real and significant. The biological mechanisms linking these conditions are multiple, converging, and increasingly well understood.
Chronic inflammation is one of the most important. Metabolic dysfunction, particularly obesity and insulin resistance, drives systemic low-grade inflammation. Inflammatory cytokines cross the blood-brain barrier and disrupt neurotransmitter systems, including serotonin and dopamine, while also impairing neuroplasticity and disrupting the HPA axis that governs the stress response. The brain in a chronically inflamed body is not operating in a neutral environment.
Insulin resistance itself has direct effects on brain function. Insulin plays a role in neuronal signaling, synaptic plasticity, and the regulation of mood-related neurotransmitters. When neurons become insulin resistant, these processes are compromised. Brain imaging studies have shown reduced glucose metabolism in depressed individuals, suggesting that the brain's energy supply is impaired in a way that mirrors what happens in peripheral metabolic dysfunction.
Mitochondrial dysfunction is another convergence point. As explored in earlier articles, mitochondria are the cellular machinery that converts metabolic inputs into usable energy. Metabolic disorders impair mitochondrial function systemically. The brain, as the most energetically demanding organ in the body, is particularly vulnerable to this impairment. Disrupted mitochondrial function in neural tissue has been documented in depression, bipolar disorder, schizophrenia, and anxiety disorders.
A horizon worth naming: the Alzheimer's connection
Perhaps the most striking frontier in the metabolic-mental health connection is the emerging understanding of Alzheimer's disease as a condition with deep metabolic roots.
A growing body of research, drawing on postmortem analyses of Alzheimer's brains, animal models, and epidemiological data, has proposed that the brain in Alzheimer's disease undergoes a form of insulin resistance that is distinct from, and in some ways more severe than, what occurs in Type 2 diabetes. This has led some researchers to use the informal term "type 3 diabetes" to describe the pattern, specifically the brain's progressive loss of the ability to respond to insulin, with corresponding impairment of glucose metabolism, neuronal energy supply, and the clearance of amyloid-beta plaques [11].
It is important to be clear about what this term does and does not mean. Type 3 diabetes is not a formally recognized diagnostic category. It has not been adopted by the WHO or the American Diabetes Association. It is a research concept, used to highlight the overlap between the metabolic mechanisms of diabetes and the neurodegeneration observed in Alzheimer's. A 2024 meta-analysis of 144 prospective studies found a 1.25 to 1.91-fold increased risk of cognitive disorders in people with diabetes [12]. A separate 2024 meta-analysis found a 59% increased risk of developing Alzheimer's in people with Type 2 diabetes [12].
The connection is compelling enough to be generating serious therapeutic interest. GLP-1 receptor agonists, the same class of drugs driving the Ozempic conversation, are now being studied for neuroprotective effects in Alzheimer's disease, with early data showing reductions in inflammatory markers and improvements in amyloid clearance in animal models. A 2025 real-world study linked GLP-1 use in people with Type 2 diabetes to a nearly 12% reduced risk of Alzheimer's disease.
We will explore the Alzheimer's-metabolic connection in depth in a dedicated future article. The science here is complex, rapidly evolving, and important enough to deserve its own careful treatment. For now, the point to hold onto is this: the relationship between metabolic dysfunction and cognitive decline appears to extend all the way from mild mood dysregulation, to clinical depression, to the neurodegenerative processes underlying dementia. This is not a peripheral concern. It may be one of the defining medical stories of the next decade.
From mild mood disruption to clinical depression to the neurodegeneration underlying Alzheimer's, metabolic dysfunction appears to run through the entire spectrum of how the brain fails. That is not a peripheral concern. It may be the story of the next decade.
Why this matters for how we think about mental health
The picture that emerges from this research challenges some of the most basic assumptions embedded in how we currently approach mental health.
If psychiatric conditions are, at least in part, metabolic conditions, then treating them exclusively with medications that target neurotransmitters, while ignoring the underlying metabolic environment, is treating symptoms rather than causes. It is the equivalent of managing the fever without addressing the infection.
This does not mean psychiatric medications are without value. For many people, they provide essential relief that makes functioning possible. But it does mean that a complete account of mental health must include the metabolic systems that the brain depends on. Insulin sensitivity, inflammation, mitochondrial function, blood sugar regulation, these are not separate from mental health. They are part of its biological foundation.
The good news embedded in this framing is that metabolic health is, to a meaningful degree, modifiable. Exercise, sleep, nutrition, stress management, and other lifestyle inputs influence the metabolic systems that underlie both physical and mental health. The research connecting these inputs to psychiatric outcomes is growing, and in some cases, already robust. Future articles will explore what that research actually shows and what it means practically.
For now, the most important thing to recognize is that the two epidemics unfolding simultaneously in America, one metabolic and one psychiatric, are not independent crises. They share a root system. Addressing them separately, as medicine has largely done, misses something fundamental about what is actually happening in the bodies and brains of tens of millions of people.
Coming up in this series
Future articles will explore the Alzheimer's-metabolic connection in depth, the bidirectional relationship between inflammation and depression, and what the research says about metabolic interventions for psychiatric conditions.
Article tags
Metabolic Dysfunction , Mental Health , Obesity , Type 2 Diabetes , Depression , Insulin Resistance , Inflammation , Alzheimer's Disease , Type 3 Diabetes , Metabolic Psychiatry , Brain Health , Psychiatric Medication
References
1. Trust for America's Health. (2024). State of Obesity 2024: Better Policies for a Healthier America. https://www.tfah.org/report-details/state-of-obesity-2024/
2. Centers for Disease Control and Prevention. (2024). National Diabetes Statistics Report. https://www.cdc.gov/diabetes/data/statistics-report/index.html
3. Translational Psychiatry. (2016). Is increased antidepressant exposure a contributory factor to the obesity pandemic? https://www.nature.com/articles/tp201625
4. Better Mind. (2025). 2025 Mental Health and Well-Being Status. https://www.bettermind.com/blog/2025-status/
5. Journal of Clinical Psychiatry / NCS-R. (2007). Association between obesity and psychiatric disorders in the US adult population. PMC. https://pmc.ncbi.nlm.nih.gov/articles/PMC1913935/
6. Endotext / NCBI Bookshelf. (2025). Diabetes and Depression. https://www.ncbi.nlm.nih.gov/books/NBK498652/
7. CDC. (2023). State-Specific Prevalence of Depression Among Adults With and Without Diabetes, United States, 2011-2019. https://www.cdc.gov/pcd/issues/2023/22_0407.htm
8. Horsbøl, T.A., et al. (2024). Diabetic complications and risk of depression and anxiety among adults with type 2 diabetes. Diabetic Medicine, 41(4), e15272. https://pubmed.ncbi.nlm.nih.gov/38157285/
9. Michigan Medicine. (2024). Mental health and chronic diabetes complications strongly linked both ways. Diabetes Care. https://www.michiganmedicine.org/health-lab/mental-health-and-chronic-diabetes-complications-strongly-linked-both-ways-study-finds
10. Centers for Disease Control and Prevention. (2024). Diabetes and Mental Health. https://www.cdc.gov/diabetes/living-with/mental-health.html
11. PMC. (2025). A systematic review on type 3 diabetes: bridging the gap between metabolic dysfunction and Alzheimer's disease. https://pmc.ncbi.nlm.nih.gov/articles/PMC12382249/
12. MDPI. (2024). Alzheimer's Disease as Type 3 Diabetes: Understanding the Link and Implications. https://www.mdpi.com/1422-0067/25/22/11955
About the Metabolic Mind Project
A psychologist-founded publication tracking the emerging science and cultural shift around metabolic approaches to mental health, before it reaches the mainstream. We publish weekly at MetabolicMindProject.com.



Comments